Electron capture decay2/11/2024 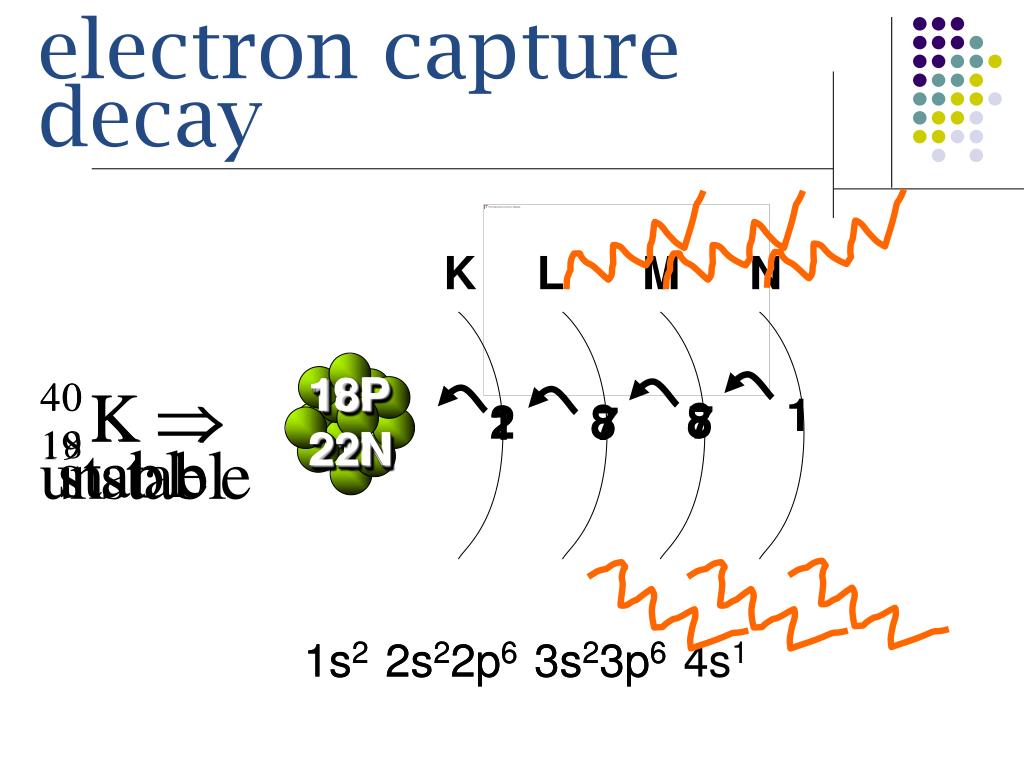
Most of the electrons orbit the nucleus at distances large compared to the nucleus. The captured electron belongs to the group of electrons orbiting around the nucleus. Ordinary beta-minus decay has no competitor on Earth however to reduce an excess of neutrons, since the capture of positrons would occur in an world made of antimatter. Electron capture, along with beta-positive decay, is Nature’s way of guaranteeing that no nucleus becomes too proton-heavy. The capture of an electron has the same effect on a nucleus as the emission of a positron: one of its protons transforms into a neutron, diminishing the global electric charge of the nucleus by 1 unit. The electron capture trigger the emission of an invisible neutrino by the nucleus. 
The best-known example is of potassium 40 : 11% of the nuclei of that isotope of potassium present in our body decay by electronic capture. In both cases, practically all the enegy released is carried by light particles.Įlectron capture is a comparatively minor decay mode caused by the weak force. There is no such energy threshold in the case of electron capture (bottom). The emission of a positron and the capture of an electron are twin reactions both resulting in the diminution of the number of protons by 1 (from Z to Z-1) and the production of a neutrino.The positron observed in the final stage of the beta decay (top) is a new particle requiring the 0.511 MeV of its rest mass energy to be created. Positron emission versus electron capture
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
